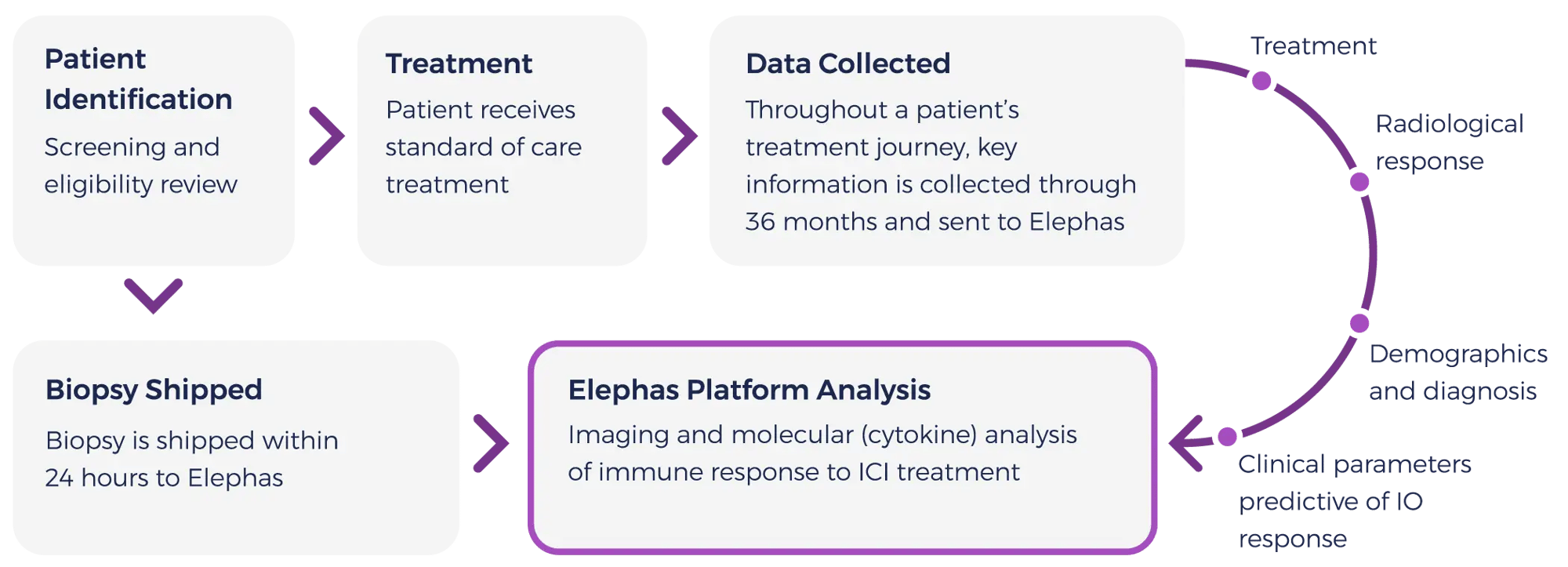

We are conducting three observational clinical trials. The primary objective of these studies is to determine the ex-vivo predictive accuracy of elive™ across a variety of solid tumors.
Study endpoints:
Measured per RECIST v1.1 and iRECIST





Observational Lung Trial to Collect Tissue to Train and Validate a Live Tumor Diagnostic Platform
Tumor types:
Non-small cell lung cancer
Setting:
Metastatic & recurrent
Complete study details and locations: NCT05478538Observational Basket Trial to Collect Tissue to Develop and Train a Live Tumor Diagnostic Platform
Tumor types:
Bladder cancerColorectal cancerEndometrial cancerHead & neck cancerKidney cancerLiver cancerMelanomaNon-small cell lung cancer
Setting:
Metastatic & recurrentAdvanced
Complete study details and locations: NCT05520099Mayo Clinic MC230901: Observational Basket Trial to Collect Tissue to Develop and Train and Validate a Live Tumor Diagnostic Platform
Tumor types:
Bladder cancerColorectal cancerEndometrial cancerHead & neck cancerKidney cancerLiver cancerMelanomaNon-small cell lung cancerTriple negative breast cancerEsophageal cancerCervical cancer
Setting:
Metastatic & recurrentAdvancedNeoadjuvantAdjuvant
Complete study details: NCT06349642These trials will provide an opportunity to demonstrate correlation between ex vivo and clinical response to immunotherapy regimens.


Hospitals and academic research centers within the US provide consented patient specimens.
If you are a physician or researcher, contact us to learn more.


